Dew point psychrometric chart10/31/2023 Along this curve, the system air is saturated with water vapor. Saturation curve This is the relative humidity equal to 100% curve. The brown diagonal lines, with values read under the temperature values on the horizontal axis moisture content, is the vertical axis, with the blue lines representing it is constant along the line. This is the green-olive colored curves, which range from 10 to 100%. Once you get that point, you gain access to several other properties (such as enthalpy) by following along the other axes. When you are given any two properties that appear on the chart, then you can find a point on the graph. The technique is the same for any combination of properties given. Given a combination of these properties, we would be expected to determine the enthalpy. The chart relates the mixture's enthalpy to T dp, T wb, h a, h r, and specific volume. Where the actual water vapor - air psychrometric chart forms a continuum of lines. But this also allows us to determine several properties for a gas-vapor mixture simultaneously. Psychrometric charts can seem more confusing because they offer several more axes on one graph instead of just two. On this page, the focus is on how to read and use the chart.Īs engineers, we have probably become comfortable with using two dimensional plots. 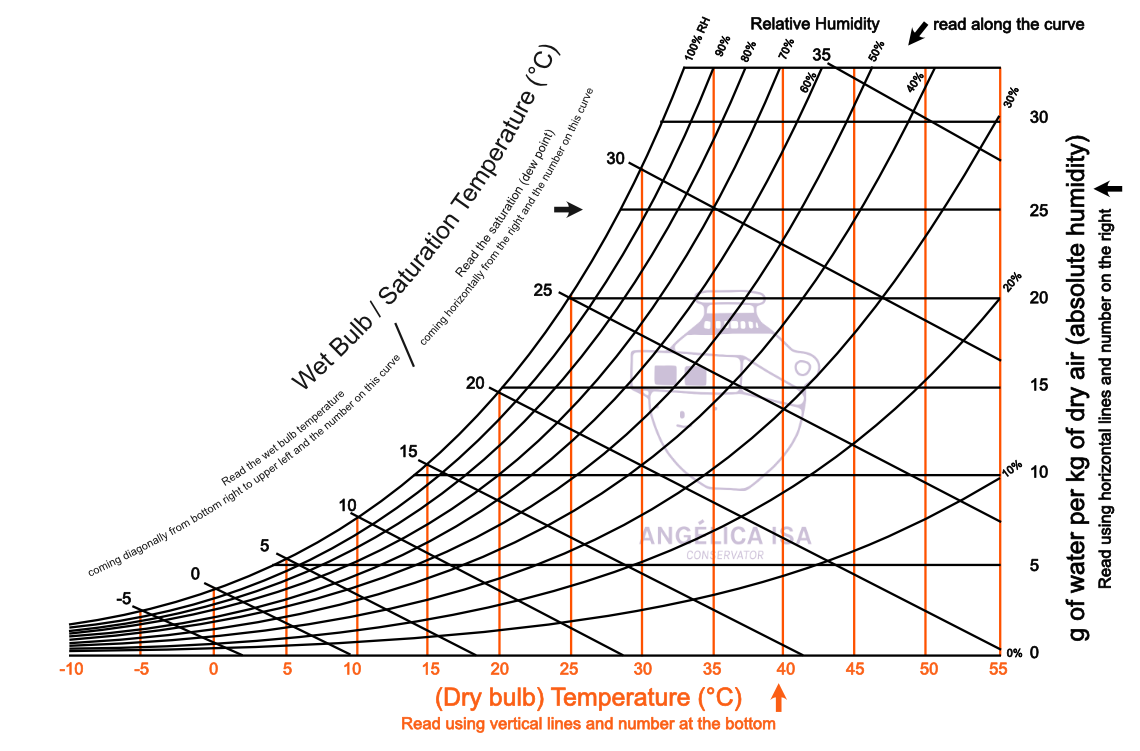
That is, it allows us to obtain enthalpies from dew point temperature and relative humidity (as an example), or from the humid specific volume and wet bulb temperature. 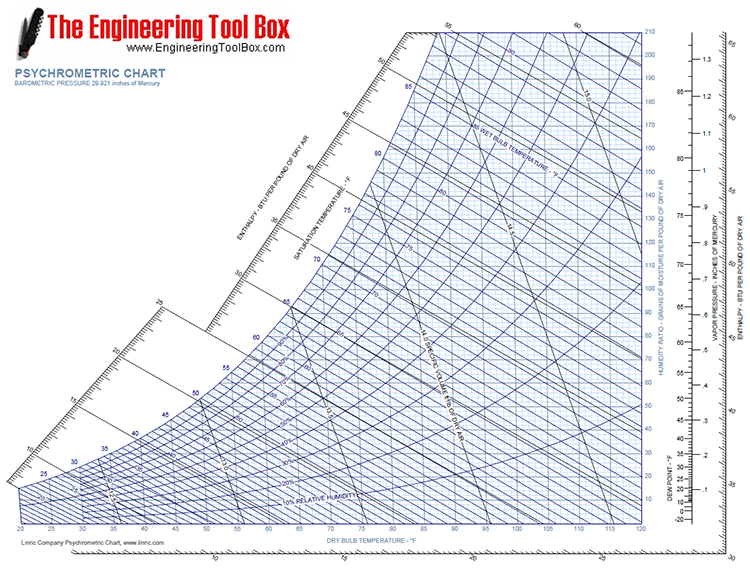
It allows us to get the enthalpies for an air-water vapor system not only directly from temperature, but also from other properties that are functions of temperature. It is just a composite of empirical correlations of different properties. However, this chart is used specifically for water vapor - air systems. From an energy balance point of view, it functions just as enthalpy tables do for obtaining enthalpies for different compounds, and as steam tables did for water vapor. Perry's Chemical Engineers' Handbook, McGraw-Hill.This page covers the use of psychrometric charts to determine the enthalpy for a water-air system. ASHRAE handbook fundamentals 2017: SI edition. Journal of Open Source Software, 4(33), 1137Īmerican Society of Heating, Refrigerating and Air-Conditioning Engineers (2017). PsychroLib: a library of psychrometric functions to calculate thermodynamic properties of air. Specific Enthalpy: Ratio of a system enthalpy to its mass.ĭ. Specific Volume: Ratio of a substance volume to its mass. Vapor Pressure: Pressure exerted by a vapor in thermodynamic equilibrium with its condensed phases at a given temperature in a closed system. At lower humidity the wet-bulb temperature is lower than dry-bulb temperature.ĭew Point Temperature: Temperature at which moisture begins to condense when mixture is cooled. At 100 % relative humidity, the wet-bulb temperature is equal to the air temperature (dry-bulb temperature). Wet Bulb Temperature: Temperature read by a thermometer covered in by a wick wetted with water which moved through air unsaturated with water vapor. Relative Humidity: Ratio of partial pressure of moisture to partial pressure of moisture at saturation.Ībsolute Humidity: Moisture content of a gas by mass. This is the temperature that is indicated by an ordinary thermometer. Reference Pressure: Default value of 101325 Pa represents the standard atmospheric pressure (1 atm).ĭry Bulb Temperature: Temperature of air measured by a thermometer freely exposed to the air.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
